It also tells you the number of in a neutral. And so, thats all going to be, as you can see, 12.01113774, which, if you were to round to the hundredths place, is how this atomic weight was gotten. 
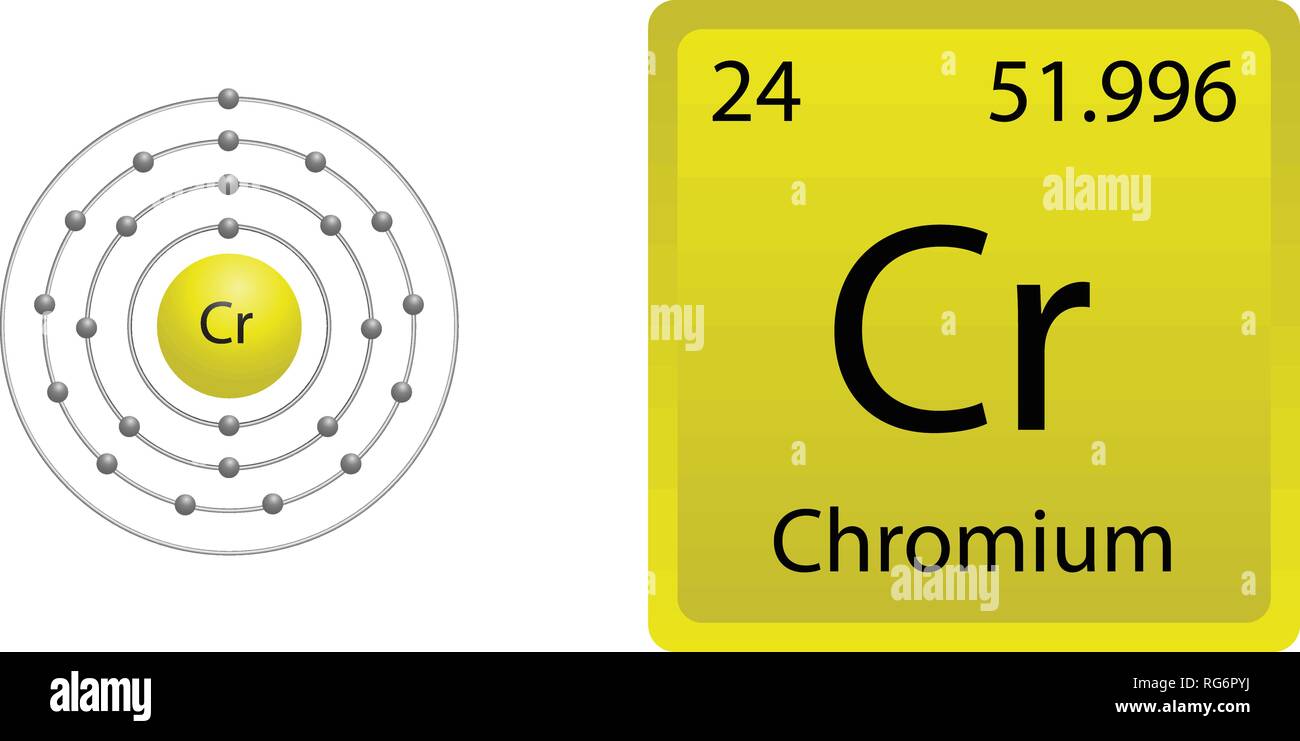
And I know its going to do this multiplication first because its a calculator knows about order of operations. From the Periodic Table we can find the element. We are going to add 0.0111 times 13.0034. For example, atoms of calcium and argon possess the same mass number which is 40u while different atomic numbers are 20 and 18 respectively. The atomic number tells you the number of in one atom of an element. In this video we’ll use the Periodic table and a few simple rules to find the protons, electrons, and neutrons for the element Chromium (Cr). Naturally occurring isotopes are marked in green light green = naturally occurring radionuclides. Atoms of different elements with different atomic numbers but the same mass number are called isobars. The mass number is used to calculate the number of. Isotones and IsobarsThe following table shows the atomic nuclei that are isotonic (same neutron number N = 34) and isobaric (same nucleon number A = 58) with Chromium-58. The number of protons in one atom of an element determines the atoms. For an isotope, the number that follows the elements name represents its mass number, which tells you how many protons and neutrons the isotope has in its nucleus. Name of the isotope: Chromium-58 Cr-58 Symbol: 58Cr Mass number A: 58 Atomic number Z: 24 (= number of protons) Neutrons N: 34 Isotopic mass: 57.9441845(16) u ( atomic weight of Chromium-58) Nuclide mass: 57.9310189 u (calculated nuclear mass without electrons) Mass excess: -51.9918 MeV Mass defect: 0.538223412 u (per nucleus) Nuclear binding energy: 501.35187894 MeV (per nucleus) 8.64399791 MeV (average binding energy per nucleon) Separation energy: S N = 7.5384(18) MeV (first neutron) S P = 14.87(80) MeV (first proton) Half-life: 7.0(3) s Decay constant λ: 0.099021025794278 s -1 Spin and parity: 0+ Year of discovery: 1980 So, you know that chromium-51 decays into manganese-51, but you dont know what type of radiation is emitted when this takes place. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
